Amer M. Johri, Marie-France Hétu, Daren K. Heyland, Julia E. Herr, Jennifer Korol, Shawna Froese, Patrick A. Norman, Andrew G. Day, Murray F. Matangi, Erin D. Michos, Stephen A. LaHaye, Fraser W. Saunders & J. David Spence
Nutrition & Metabolism volume 19, Article number: 26 (2022) Cite this article
Abstract
Background
L-carnitine (L-C), a ubiquitous nutritional supplement, has been investigated as a potential therapy for cardiovascular disease, but its effects on human atherosclerosis are unknown. Clinical studies suggest improvement of some cardiovascular risk factors, whereas others show increased plasma levels of pro-atherogenic trimethylamine N-oxide. The primary aim was to determine whether L-C therapy led to progression or regression of carotid total plaque volume (TPV) in participants with metabolic syndrome (MetS).
Methods
This was a phase 2, prospective, double blinded, randomized, placebo-controlled, two-center trial. MetS was defined as ≥ 3/5 cardiac risk factors: elevated waist circumference; elevated triglycerides; reduced HDL-cholesterol; elevated blood pressure; elevated glucose or HbA1c; or on treatment. Participants with a baseline TPV ≥ 50 mm3 were randomized to placebo or 2 g L-C daily for 6 months.
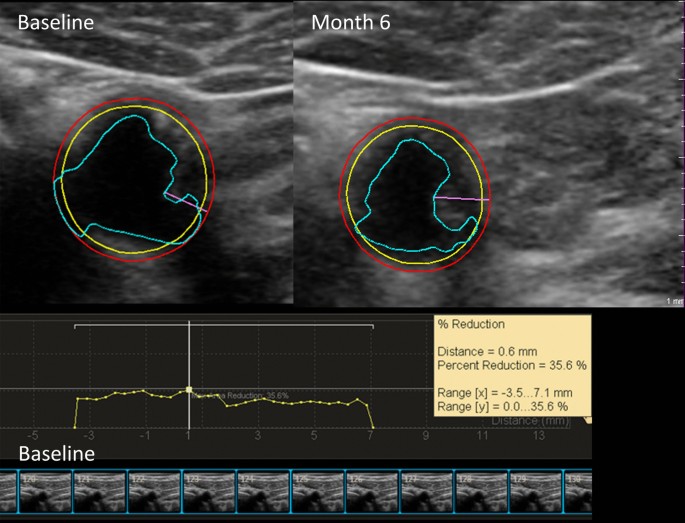
Results
The primary outcome was the percent change in TPV over 6 months. In 157 participants (L-C N = 76, placebo N = 81), no difference in TPV change between arms was found. The L-C group had a greater increase in carotid atherosclerotic stenosis of 9.3% (p = 0.02) than the placebo group. There was a greater increase in total cholesterol and LDL-C levels in the L-C arm.
Conclusions
Though total carotid plaque volume did not change in MetS participants taking L-C over 6-months, there was a concerning progression of carotid plaque stenosis. The potential harm of L-C in MetS and its association with pro-atherogenic metabolites raises concerns for its further use as a potential therapy and its widespread availability as a nutritional supplement.
Trial registration: ClinicalTrials.gov, NCT02117661, Registered April 21, 2014, https://clinicaltrials.gov/ct2/show/NCT02117661.
https://nutritionandmetabolism.biomedcentral.com/articles/10.1186/s12986-022-00661-9